An Isotope With 27 Protons and 32 Neutrons
Cobalt-60 is composed of 27 protons 33 neutrons and 27 electrons. Constants Period Part A an isotope with 27 protons and 32.
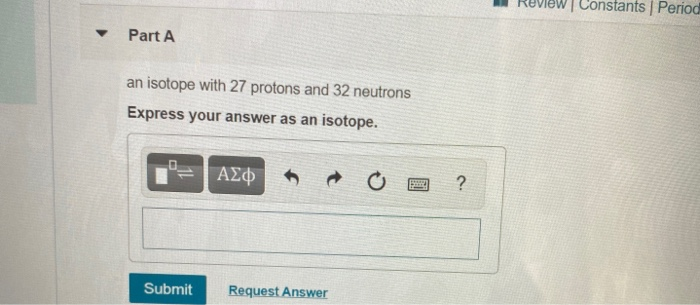
Solved Constants Period Part A An Isotope With 27 Protons Chegg Com
The molar mass of aluminium which is average of atomic masses of all isotopes 26981538 gmol since 13 Al 27 is the major isotope.
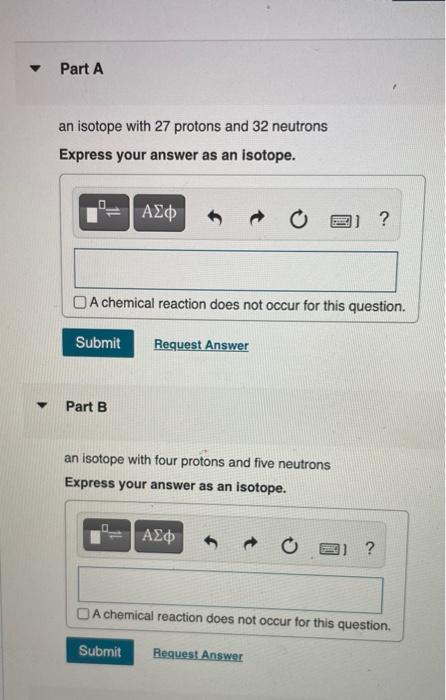
. 8 protons 8 neutrons 8 electrons. B The nucleus comprises a very small fraction of the total volume of the atom. Knowing that the element have 26 protons and 26 electrons it have to be an isotope of iron.
Youll probably want a periodic table handy. The number of neutrons is given by subtracting the atomic mass from the proton number. Element j has 27 protons and 32 neutrons element l has 27 protons and 33 neutrons isotope element x has 17 protons and 18 neutrons element y has 18 protons and 17 neutrons.
How many protons does uranium have. Protons- 47 Neutrons- 60 Electrons- 47. Protons- 79 Neutrons- 118 Electrons- 79.
Firstly cobalt-60 is an isotope. An atom with 14 electrons and 15 neutrons 4. How are the number of protons neutrons and electrons in an element.
59 Co is the only stable cobalt isotope and the only isotope that exists naturally on Earth. UraniumAtomic number Uranium is a silvery-white metallic chemical element in the periodic table with atomic number 92. Isotope Practice of protons neutrons electrons STUDY.
C The number of neutrons in the nucleus equals the number of protons. Protons- 20 Neutrons- 20 Electrons- 20. 112 rows 27.
Protons- 1 Neutrons- 1 Electrons- 1. So we have 42 protons and if its neutral we have 42 electrons and then well have 96 minus 42 or 54 neutrons. To get 32 neutrons from a lived in um 96 96 is the mass number.
Name how many protons neutrons and electrons each isotope has. 27 protons 32 neutrons 27 electrons. The number neutrons isotope atomFAQhow scientists determine the number neutrons isotope atomadminSend emailNovember 28 2021 minutes read You are watching how scientists determine the number.
15 protons 16 neutrons 15 electrons. Terms in this set 21 Hydrogen-2. Protons- 12 Neutrons- 16 Electrons- 12.
E The protons and neutrons together comprise most of the mass of the. Mass number of the isotope number of protons number of neutrons. We need to look up the atomic number from the periodic table which is 42.
Write the atomic symbols for isotopes with the following. To determine the number of neutrons we will take the 59 minus the number of protons 27. 100 1 rating Transcribed image text.
Its mass number is 56. Since the sum of the number of protons and neutrons in your particular question is 59 the answer to what has 27 protons and 32 neutrons is. A neon atom with 12 neutrons 3.
1 proton 1 neutron 1 electron. What element has a mass number of 27 and 14 neutrons the number of neutrons in above isotope A Z 27 13 14. Correct answer - An isotope with 27 protons and 32 neutrons Express your answer as an isotope.
Protons- 27 Neutrons- 32 Electrons- 27. A nickel atom with 32 neutrons Express your answer as. Protons- 27 Neutrons- 32 Electrons- 27.
A mass number of 24 and 13 neutrons 5. So in the case of 40 18Ar then it has 18 protons and 18 electrons. Name how many protons neutrons and electrons each isotope has.
An isotope of iron Fe has 26 protons and 32 neutrons. Protons- 1 Neutrons- 1 Electrons- 1. And since you are inquiring about the isotope Cobalt 60 then it has 60 -.
In the form of an element Y ZX Z is the number of protons and also the number of electrons and Y is the atomic mass. The most stable 60 Co has a half-life of 52714 years. 16 protons 18 neutrons 16 electrons.
Twenty-two radioisotopes have been characterized. Amu - atomic mass units. We review their content and use your feedback to keep the quality high.
Cobalt-59 is composed of 27 protons 32 neutrons and 27 electrons. D The electrons comprise the nucleus of the atom. A The number of protons equals the number of electrons in a neutral atom.
6 protons 6 protons 6 protons. Cobalt has 27 protons 32 neutrons and 27 electrons. Experts are tested by Chegg as specialists in their subject area.
Mass number of the isotope 26 32 58 amu. What is the mass number of element A if the proton is 26 electron is 26 and the neutron is 32. Cobalt is element 27 so it has 27 protons.
An atom with 27 protons and 32 neutrons 2. Terms in this set 15 Hydrogen-2. Nickel has 28 protons 31.
Diagram of the nuclear composition electron configuration chemical data and. Youll probably want a periodic table handy.
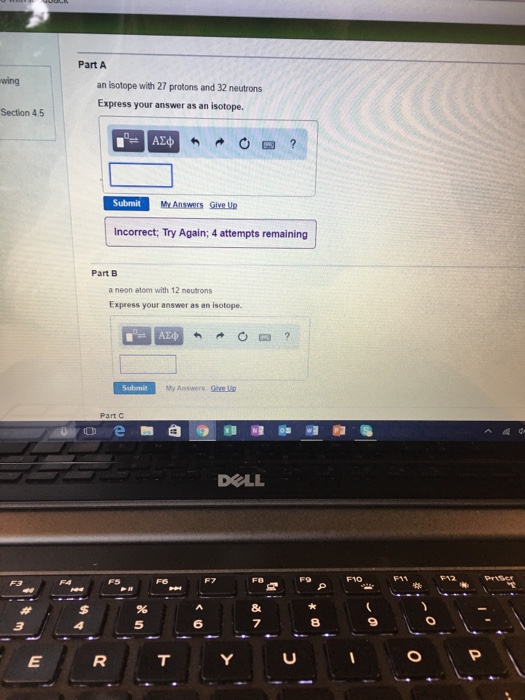
Solved Part A Wing An Lsotope With 27 Protons And 32 Chegg Com
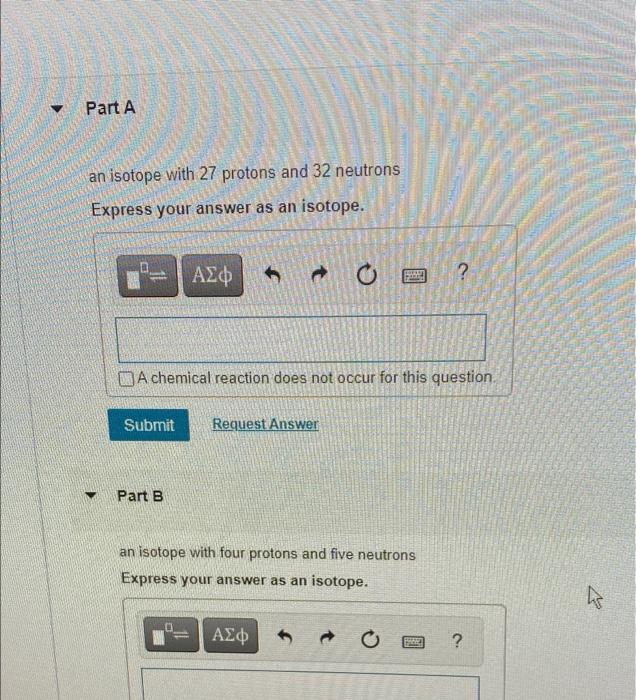
Solved Part A An Isotope With 27 Protons And 32 Neutrons Chegg Com

Solved Part A An Isotope With 27 Protons And 32 Neutrons Chegg Com
Comments
Post a Comment